Why in news?
On 27 February 1940, chemists Martin Kamen and Samuel Ruben discovered the radioactive isotope carbon‑14 at the University of California, Berkeley. Live Science revisited this milestone in February 2026, highlighting how the discovery revolutionised archaeology and other fields.
Background
Scientists had hypothesised the existence of a “heavy” carbon isotope with two extra neutrons but assumed it would be short‑lived. Ernest Lawrence tasked Kamen and Ruben with producing it using a cyclotron. In January 1940, after many failed attempts, they bombarded graphite with deuterons (heavy hydrogen nuclei) for 120 hours. The result was a radioactive form of carbon with a long half‑life.
Key aspects of carbon‑14
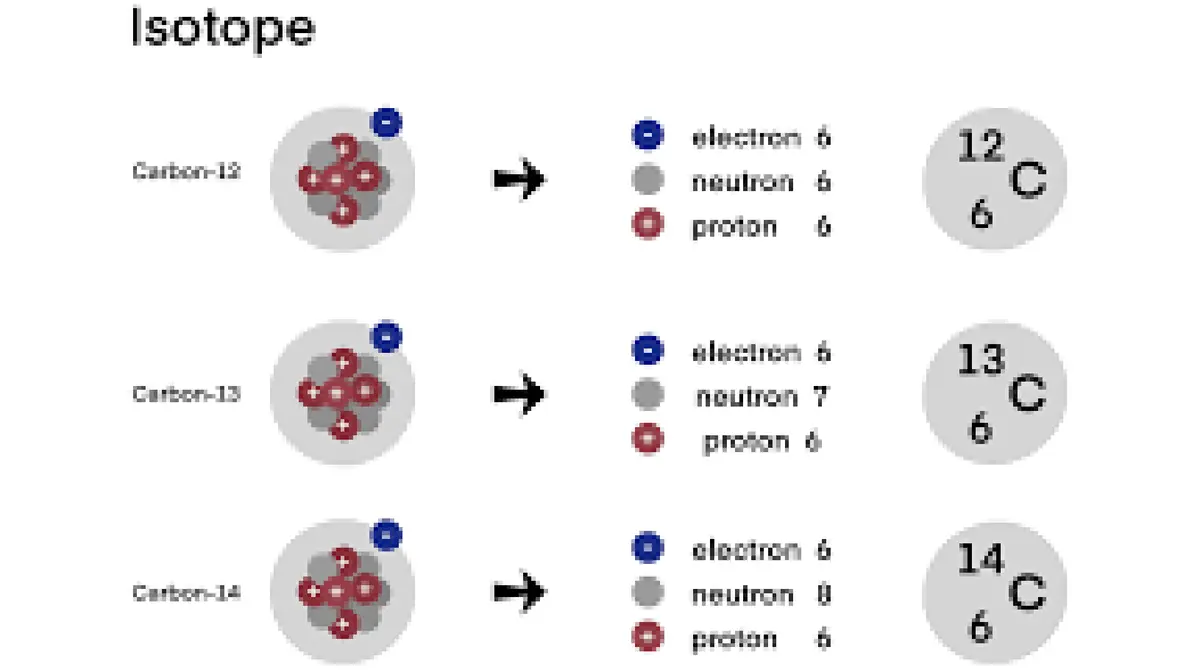
- Radioactive nature: Carbon‑14 has six protons and eight neutrons. It forms naturally in Earth’s upper atmosphere when cosmic rays strike nitrogen atoms. Its half‑life of about 5,730 years means it decays slowly to nitrogen‑14.
- Radiocarbon dating: In 1949 chemists James Arnold and Willard Libby demonstrated that the ratio of carbon‑14 to stable carbon (carbon‑12 and carbon‑13) in organic materials can be used to estimate their age. Living organisms absorb carbon‑14 while alive; after death, the isotope decays at a predictable rate. Libby received the 1960 Nobel Prize in Chemistry for this work.
- Applications: Radiocarbon dating is now widely used in archaeology, geology, paleoclimatology and even biomedical research to date samples up to about 50,000 years old. It has helped determine the ages of prehistoric artefacts, fossils, ice cores and ancient artworks.
Significance
- Window into the past: Radiocarbon dating allows scientists to place archaeological finds in chronological context, transforming the study of human history.
- Cross‑disciplinary impact: Techniques inspired by carbon‑14 decay have been developed for other radioactive isotopes, enabling studies in earth sciences and medicine.
- Historical narrative: The story of Kamen and Ruben reflects scientific perseverance; both scientists later faced personal challenges, and their discovery paved the way for future breakthroughs.
Source: Live Science