Why in news?
A biotechnology company, PRG S&T, recently signed a licensing agreement with Sentynl Therapeutics (a Zydus Group company) to develop and commercialise Progerinin, a first‑in‑class drug candidate for treating Hutchinson–Gilford Progeria Syndrome. The agreement, announced in March 2026, brings hope for patients with this ultra‑rare genetic disorder.
Background
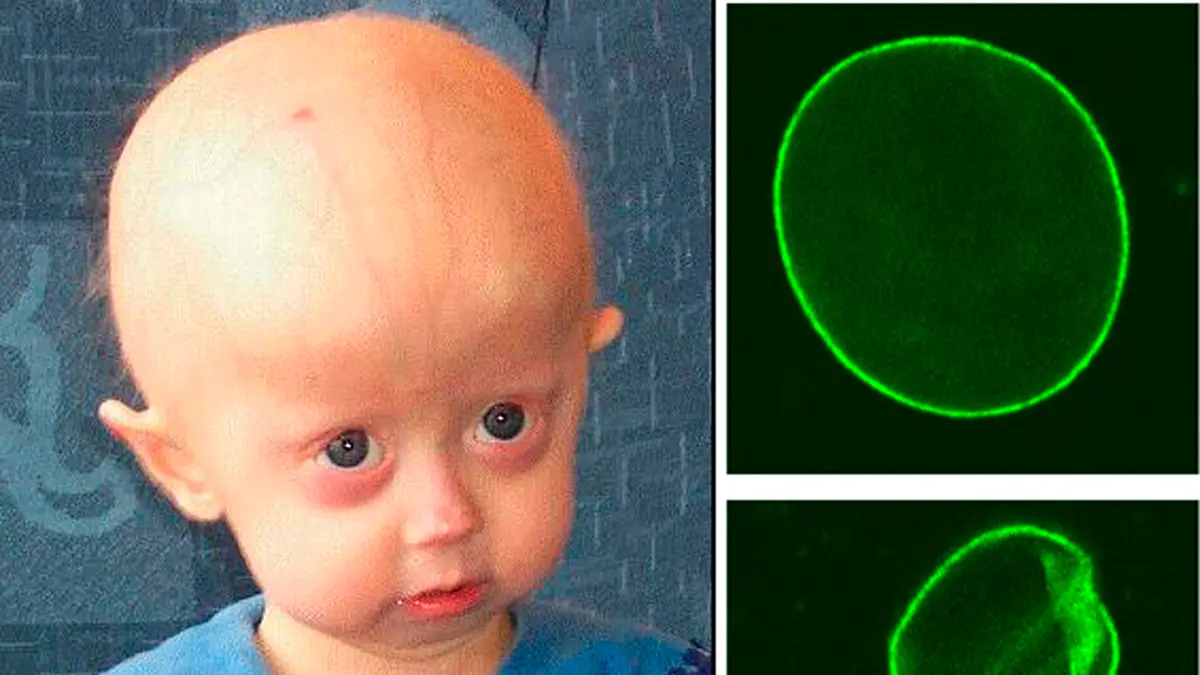
Hutchinson–Gilford Progeria Syndrome is a rare genetic disease that causes children to age rapidly. It is caused by a mutation in the LMNA gene, which leads to production of an abnormal protein called progerin. Progerin damages the structural support of the cell nucleus, leading to features such as hair loss, wrinkled skin, stiffness of joints and a beaked nose. Symptoms usually appear in the first two years of life, and affected children have an average life expectancy of about 14½ years. The condition is not inherited; it usually arises from a new mutation during conception. There is currently no cure, though the drug Lonafarnib can slow disease progression and extend life by a few years.
Progerinin and recent developments
- Mechanism of action: Progerinin is designed to reduce levels of the toxic progerin protein and restore the normal structure of the nuclear envelope. In pre‑clinical tests on mouse models, it prolonged survival from an average of 16.8 weeks to about 25.2 weeks.
- Regulatory designations: the drug has received Rare Pediatric Disease and Orphan Drug designations from regulators, indicating its potential for an underserved patient group.
- Clinical trials: Phase 2a trials began in January 2025, and dosing was expected to be completed by March 2026. The licensing deal allows Sentynl to commercialise the drug globally after successful trials.
Importance
Progeria affects around 1 in 18 million people worldwide, and options are extremely limited. The development of Progerinin could provide a more effective therapy by targeting the root cause of the disease rather than managing symptoms. If successful, it may improve quality of life and extend survival for children with HGPS.
Source: News Bytes