Why in news?
Geneticists and clinicians are increasingly advocating whole‑exome sequencing as a first‑line test for diagnosing rare genetic disorders. In April 2026 new clinical guidelines highlighted that examining only the protein‑coding portion of the genome can provide faster and more cost‑effective answers for patients with unexplained developmental delays or congenital anomalies.
Background
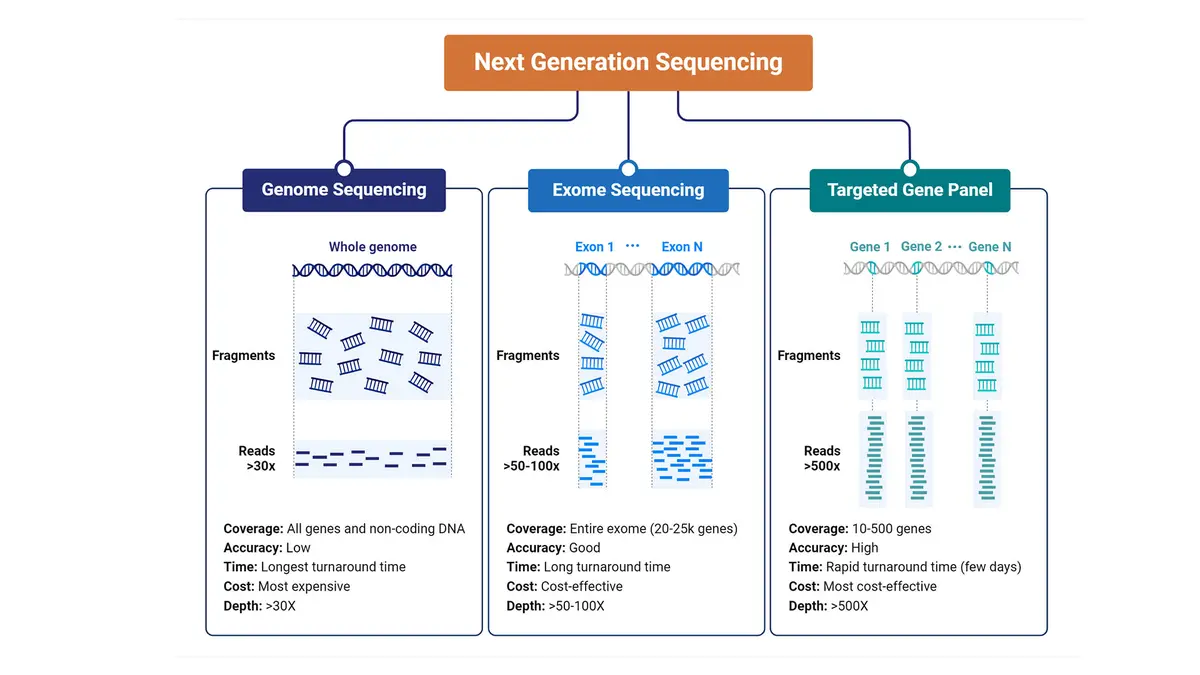
The human genome contains about 20 000 protein‑coding genes, whose exons constitute less than two percent of our DNA. Yet more than 80 percent of disease‑causing variants lie in these regions. Whole‑exome sequencing (WES) captures and sequences only these coding segments. Compared with whole‑genome sequencing (WGS), WES generates far less data, making analysis quicker and more affordable while still covering most known pathogenic variants. The process involves enriching exons from a patient’s DNA, sequencing them on a high‑throughput platform and comparing the results against reference genomes to identify rare or deleterious changes.
Applications and advantages
- Diagnostic yield: WES can provide a genetic diagnosis for roughly 20–50 percent of patients with suspected monogenic disorders, outperforming targeted gene panels or chromosomal microarray tests.
- Cost‑effectiveness: By focusing on the two percent of the genome where most actionable variants reside, WES reduces sequencing costs and data‑analysis burdens compared with whole‑genome sequencing.
- Broad utility: It is particularly useful for genetically heterogeneous conditions such as developmental delay, intellectual disability, epilepsy and congenital anomalies, where many different genes can cause similar symptoms.
- Research tool: WES helps researchers discover new disease genes and understand how mutations affect protein function, contributing to the development of targeted therapies.
Limitations
- Incomplete coverage: WES does not examine regulatory or intronic regions of the genome and may miss structural rearrangements or non‑coding mutations that affect gene expression.
- Variant interpretation: Many detected changes are of uncertain significance and require clinical correlation and functional studies to determine whether they cause disease.
- Ethical considerations: Sequencing can uncover incidental findings unrelated to the patient’s current condition, raising questions about consent and counselling.
Conclusion
Whole‑exome sequencing is transforming medical genetics by making it feasible to search across thousands of genes at once. Used judiciously alongside clinical assessment, it can shorten diagnostic journeys and inform personalised treatment strategies.